Publication
589
J. Am.
Chem. Soc, 127 (14), 5049-5055,
2005
DOI: 10.1021/ja042940f S0002-7863(04)02940-3
|
|
|
Role of protonation and of axial ligands in the reductive
dechlorination of alkyl chlorides by
vitamin B12 complexes. Reductive cleavage
of chloroacetonitrile by Co(I) cobalamins
and cobinamids. |
|
|
|
| |
 |
|
|
|
|
|
|
Juan E. Argüello, Cyrille Costentin, Sophie Griveau, and Jean-Michel Savéant
Contribution from the Laboratoire
d'Electrochimie Moléculaire,
UMR 7591, Université Paris 7-CNRS, 2 place Jussieu, F-75251 Paris Cedex
05, France
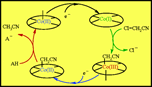
Cobalt(I) cobalamin and cobinamide are efficient catalysts of the
hydrogenolysis of aliphatic chloro compounds. Taking chloroacetonitrile as
example, the first requirement for high catalytic efficiency is fulfilled by the
high reactivity of the Co(I) complex toward the substrate, leading to the
alkylcobalt(III) complex. This is further reduced into the alkylcobalt(II)
complex. However, the fact that these two reactions are fast is not enough to
ensure an efficient catalysis: in DMF catalysis is very poor, while it is high
in water. The experiments carried out in DMF with addition of an acid show that
a crucial step in the catalytic process is the proton transfer decomposition of
the alkylcobalt(II) complex, leading to the product, and closing the catalytic
loop by regeneration of the cobalt(I) complex. Another important feature of
these catalytic reactions is the role played by axial ligands present in the
solution, particularly those that are produced by the catalytic reaction itself,
namely, chloride ions and the counteranion of the added acid. The stronger these
ligands, the more negative the potential required for the reduction of the
alkylcobalt(III) complex. This amounts to a self-moderation effect: the more
efficient catalysis, the slower its second step, i.e., the conversion of the
alkylcobalt(III) complex into the alkylcobalt(II). |

