Publication
619
J. Am. Chem. Soc , 129, 9201-9209, 2007.
DOI:
10.1021/ja0714787
|
|
|
|
|
|
 |
Electrochemical Study of a Reconstituted Photosynthetic Electron-Transfer Chain |
|
|
|
|
|
|
|
Vincent Fourmond, Bernard Lagoutte, Pierre Sétif, Winfried Leibl * and Christophe Demaille **
*Contribution from the CEA, Institut de Biologie et de Technologies de Saclay, URA 2096, Gif sur Yvette, F-91191, France, and ** Laboratoire d'Électrochimie Moléculaire, UMR du CNRS No7591, Université Paris VII-Denis Diderot, 75251 Paris cedex 05, France
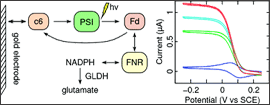
A multi-enzyme electron-transfer chain involving solubilized photosystem I (PSI) as photocatalytic unit, cytochrome c6 and ferredoxin as electron carriers and ferredoxin/NADPH oxidoreductase (FNR) as electron acceptor was reconstituted in an electrochemical cell and studied by cyclic voltammetry. The working gold electrodes were modified to react selectively with cytochrome c6. Quantitative analysis of the photocatalytic current under continuous illumination allowed the determination of the values kon and koff for the ferredoxin/PSI interaction. An efficient recycling system for NADPH was established, and the dissociation constant of the oxidized ferredoxin/semiquinone FNR complex was extracted by modeling the catalytic efficiency of the chain as a function of ferredoxin concentration. The value determined hereby is consistent with a shift of -50 to -100 mV of the reduction potential of ferredoxin when complexed with FNR |

