Publication
665
J. Am. Chem. Soc. ,132 (29),
10142 -10147, 2010.
DOI: 10.1021/ja103421f
|
|
|
|
|
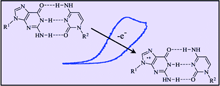 |
Effect of base pairing on the electrochemical oxidation of guanine |
|
|
|
|
|
Cyrille Costentin, Viviane Hajj, Marc Robert, Jean-Michel Savéant and Cédric Tard
Laboratoire d'Electrochimie Moléculaire, Université Paris Diderot, UMR CNRS 7591, 15, rue Jean-Antoine de Baïf, 75205 Paris Cedex 13, France
The effect of base pairing by cytosine on the electrochemical oxidation of guanine is examined by means of cyclic voltammetry on carefully purified reactants in a solvent, CHCl3, which strongly favors the formation of an H-bonded pair. The thermodynamics and kinetics of the oxidation reaction are not strongly influenced by the formation of the pair. They are actually similar to those of the reaction in which 2,6-lutidine, an encumbered base that cannot form a pair with guanine, replaces cytosine. The reaction does not entail a concerted proton−electron mechanism, as attested by the absence of H/D isotope effect. It rather involves the rate-determining formation of the cation radical, followed by its deprotonation and dimerization of the resulting neutral radical in competition with its further oxidation. |

