Publication
680
Journal of Molecular Structure, 990, 32-36, 2011
DOI: 10.1016/j.molstruc.2010.12.050
|
|
|
|
|
 |
σ-Hole bonding in 15N-labeled N-Benzyl-N-(4-iodo-tetraflurobenzyl)-amine: Synthesis, crystal structure and solid-state structure calculations |
|
|
|
|
Noureddine Raouafi, Peter Mayer , Khaled Boujlel, Bernd Schöllhorn
Laboratoire de Chimie Analytique et d’Electrochimie, Département de Chimie, Faculté des Sciences de Tunis, Université El-Manar, Rue Béchir Salem Belkhiria, 2092 Tunis El-Manar, Tunisia - Department Chemie und Biochemie, Ludwig-Maximilians-Universität München, Butenandtstr. 5-13, Haus D, 81377 München, Germany - Laboratoire d’Electrochimie Moléculaire, UMR CNRS 7591, Université Paris Diderot, 15 rue Jean-Antoine de Baïf, 75205 Paris Cedex 13, France
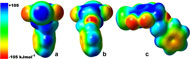
Reductive amination of 4-iodo-tetrafluorobenzaldehyde 2 and 15N-enriched benzylamine yielded the title compound 1. Single crystal X-ray diffraction (XRD) revealed that the product crystallizes in the triclinic system of the P-1 space group. The structure is consisting of infinite one-dimensional chair like chains, based on intermolecular N···I halogen bonding. Only intermolecular weak hydrogen bonds N—H···F and C—H···F are observed. Representative XRD data have been compared to the results of theoretical semi-empirical calculations in the solid-state obtained using the PM6 method. Charges of I, N and F atoms are calculated from Natural Bond Orbital (NBO) and Electrostatic Potential Surface maps have been estimated by applying second-order Møller–Plesset (MP2) perturbation theory, and confirmed clearly the assumption of σ-hole bonding formation |

