Publication
690
J. Am. Chem. Soc. 133 (32), 12801-12809, 2011
DOI:10.1021/ja204637d
|
|
|
|
|
 |
Effect of Substrate Inhibition and Cooperativity on the Electrochemical Responses of Glucose Dehydrogenase. Kinetic Characterization of Wild and Mutant Types |
|
|
|
|
Fabien Durand, Benoît Limoges, Nicolas Mano, François Mavré, Rebeca Miranda-Castro, and Jean-Michel Savéant
Laboratoire d’Electrochimie Moléculaire, UMR 7591 CNRS, Université Paris Diderot, Sorbonne Paris Cité, 15 rue Jean-Antoine de Baïf, F-75205 Paris Cedex 13, France
Centre de Recherche Paul Pascal, Universite de Bordeaux, UPR 8641, Avenue Albert Schweitzer, 33600 Pessac, France
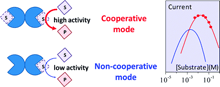
Thanks to its insensitivity to dioxygen and to its good catalytic reactivity, and in spite of its poor substrate selectivity, quinoprotein glucose dehydrogenase (PQQ-GDH) plays a prominent role among the redox enzymes that can be used for analytical purposes, such as glucose detection, enzyme-based bioaffinity assays, and the design of biofuel cells. A detailed kinetic analysis of the electrochemical catalytic responses, leading to an unambiguous characterization of each individual steps, seems a priori intractable in view of the interference, on top of the usual ping–pong mechanism, of substrate inhibition and of cooperativity effects between the two identical subunits of the enzyme. Based on simplifications suggested by extended knowledge previously acquired by standard homogeneous kinetics, it is shown that analysis of the catalytic responses obtained by means of electrochemical nondestructive techniques, such as cyclic voltammetry, with ferrocene methanol as a mediator, does allow a full characterization of all individual steps of the catalytic reaction, including substrate inhibition and cooperativity and, thus, allows to decipher the reason that makes the enzyme more efficient when the neighboring subunit is filled with a glucose molecule. As a first practical illustration of this electrochemical approach, comparison of the native enzyme responses with those of a mutant (in which the asparagine amino acid in position 428 has been replaced by a cysteine residue) allowed identification of the elementary steps that makes the mutant type more efficient than the wild type when cooperativity between the two subunits takes place, which is observed at large mediator and substrate concentrations. A route is thus opened to structure–reactivity relationships and therefore to mutagenesis strategies aiming at better performances in terms of catalytic responses and/or substrate selectivity. |

