Publication
736
J. Nanopart. Res., 15, 1-10, 2013
DOI:10.1007/s11051-013-2140-1
|
 PDF PDF |
|
|
|
|
|
|
Synergetic effect of CdS quantum dots and TiO2 nanofibers for photoelectrochemical hydrogen generation. |
|
|
|
S. Chaguetmi, F. Mammeri, M. Pasut, S. Nowak, H. Lecoq, P. Decorse, C. Costentin, S. Achour and S. Ammar
Interfaces, Traitements, Organisation et Dynamique des Systèmes, Université Paris Diderot, CNRS UMR7086, Sorbonne Paris Cité, 15 rue Jean-Antoine de Baïf, Paris Cedex 13, France
Université 20 Août 1955 de Skikda, Skikda, Algeria
Laboratoire de Céramique, Université de Constantine 1, Constantine, Algeria
Université Paris Diderot, Sorbonne Paris Cité, Laboratoire d’Electrochimie Moléculaire, Unité Mixte de Recherche Université-CNRS no. 7591, Bâtiment Lavoisier, 15 rue Jean de Baı̈f, 75205 Paris Cedex 13, France
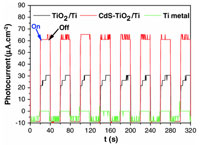
In this work, we developed a new type of nanostructured photoanodes for photoelectrochemical water splitting. They are based on CdS–TiO2 nanocomposite films, supported on conductive Ti sheets, prepared by an easy-to-achieve three-step method. It involves the production of TiO2 nanofibers (NFs) using a controlled corrosion route of polished Ti sheets, the preparation of size-controlled CdS quantum dots (QDs) by the polyol process and the direct impregnation of TiO2/Ti sheets by QDs in suspension. The photoelectrochemical (PEC) properties of the resulting nanostructures were measured, using a homemade electrochemical cell illuminated with a standard Xenon lamp, and compared to those of bare TiO2 NFs. A net enhancement of the photocurrent was observed after CdS impregnation, suggesting a low carrier recombination rate and a higher efficiency of the PEC device for solar water splitting, as the induced photocurrent is related to the electrons needed to reduce H+ ions into H2 at the cathode electrode (Pt wire). |

