Publication
747
Chem. Sci., 5 (1), 2304-2310, 2014
DOI:10.1039/C3SC53469C
|
|
|
|
|
|
|
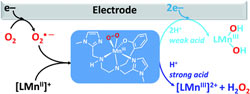
Electrochemical formation and reactivity of a manganese peroxo complex: acid driven H2O2 generation vs. O-O bond cleavage. |
|
|
|
Vincent Ching, Elodie Anxolabéhère-Mallart, Hannah E. Colmer, Cyrille Costentin, Pierre Dorlet, Timothy A. Jackson, Clotilde Policar, and Marc Robert
Univ Paris Diderot, Sorbonne Paris Cité, Laboratoire d'Electrochimie Moléculaire, UMR 7591 CNRS, 15 rue Jean-Antoine de Baïf, F-75205 Paris Cedex 13, France
Laboratoire des BioMolécules, CNRS UMR7203, Université Pierre et Marie Curie, Département de Chimie de l'ENS, 24, rue Lhomond, 75005 Paris, France
The University of Kansas, Chemistry Department, 1251 Wescoe Hall Drive, Lawrence, KS 66045, USA
CNRS, Laboratoire Stress Oxydant et Détoxication, UMR 8221, Bât 532 CEA Saclay, 91191 Gif-sur-Yvette Cedex, France
The formation of a side-on peroxo [MnIIIL(O2)] complex (L = phenolato-containing pentadentate ligand), resulting from the reaction of electrochemically reduced O2 and [MnIIL]+, is monitored in DMF using cyclic voltammetry, low temperature electronic absorption spectroscopy and electron paramagnetic resonance spectroscopy. Mechanistic studies based on cyclic voltammetry reveal that upon addition of a strong acid the Mn–O bond is broken, resulting in the release of H2O2, whereas in the presence of a weak acid the O–O bond is cleaved via a concerted dissociative electron transfer. This dichotomy of M–O versus O–O bond cleavage is unprecedented for peroxomanganese(III) complexes and the latter offers a route for electrochemical O2 activation by a manganese(II) complex. |

