Publication
754
Org. Biomol. Chem., 12, 5360-5364, 2014
DOI:10.1039/C4OB00868E
|
|
|
|
|
|
|
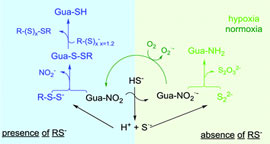
Electrophilic sulfhydration of 8-nitro-cGMP involves sulfane sulfur. |
|
|
|
V. Terzi, D. Padovani, V. Balland, I. Artaud, and E. Galardon
UMR 8601, LCBPT, CNRS-Université Paris Descartes, Sorbonne Paris Cité, 45 rue des Sts Pères, 75006 Paris, France
Laboratoire d'Electrochimie Moléculaire, UMR CNRS 7591, Université Paris Diderot, Sorbonne Paris Cite, 15 rue J.-A. de Baïf, 75205 Paris, France
The formation of 8-SH-cGMP from the reaction between hydrogen sulfide and 8-nitro-guanosine-3´,5´-cyclic monophosphate in the presence of thiols does not take place by nucleophilic attack of the hydrosulfide anion, as previously proposed, but first involves the formation of reactive species containing sulfan sulfur, like persulfides. |

