Publication
755
J. Am. Chem. Soc., 136 (33), 11821-11829, 2014
DOI:10.1021/ja506193v
|
|
|
|
|
|
|
|
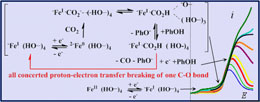
Pendant Acid–Base Groups in Molecular Catalysts: H-Bond Promoters or Proton Relays? Mechanisms of the Conversion of CO2 to CO by Electrogenerated Iron(0)Porphyrins Bearing Prepositioned Phenol Functionalities. |
|
|
|
Cyrille Costentin, Guillaume Passard, Marc Robert, and Jean-Michel Savéant
Université Paris Diderot, Sorbonne Paris Cité, Laboratoire d’Electrochimie Moléculaire, Unité Mixte de Recherche Université - CNRS No 7591, Bâtiment Lavoisier, 15 rue Jean de Baïf, 75205 Paris Cedex 13, France
Two derivatives of iron tetraphenylporphyrin bearing prepositioned phenolic functionalities on two of the opposed phenyl groups prove to be remarkable catalysts for the reduction of CO2 to CO when generated electrochemically at the Fe0 oxidation state. In one case, the same substituents are present on the two other phenyls, whereas in the other the two other phenyls are perfluorinated. They are taken as examples of the possible role of pendant acid–base groups in molecular catalysis. The prepositioned phenol groups incorporated into the catalyst molecule induce strong stabilization of the initial Fe0CO2 adduct through H-bonding, confirmed by DFT calculations. This positive factor is partly counterbalanced by the necessity, resulting from the same stabilization, to inject an additional electron to trigger catalysis. Thanks to the preprotonation of the initial Fe0CO2 adduct, the potential required for this second electron transfer is not very distant from the potential at which the adduct is generated by addition of CO2 to the Fe0 complex. The protonation step involves an internal phenolic group and the reprotonation of the phenoxide ion thus generated by added phenol. The prepositioned phenol groups thus play both the role of H-bonding stabilizers and high-concentration proton donors. They play the same role in the second electron transfer step which closes the catalytic loop concertedly with the breaking of one of the two C–O bonds of CO2 and with proton transfer. It is also remarkable that reprotonation by added phenol is concerted with the three other events. |

