Publication
828
J. Am. Chem. Soc., 139 (24), 8245-8250, 2017
DOI:10.1021/jacs.7b02879
|
|
|
|
|
|
 |
Homogeneous Molecular Catalysis of Electrochemical Reactions: Catalyst Benchmarking and Optimization Strategies
|
|
|
|
Cyrille Costentin, and Jean-Michel Savéant
Université Paris Diderot, Sorbonne Paris Cité, Laboratoire d’Electrochimie Moléculaire, Unité Mixte de Recherche Université—CNRS No. 7591, Bâtiment Lavoisier, 15 rue Jean de Baïf, 75205 Paris Cedex 13, France
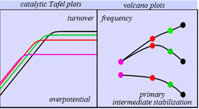
Modern energy challenges currently trigger an intense interest in catalysis of redox reactions—electrochemical and photochemical—particularly those involving small molecules such as water, hydrogen, oxygen, proton, carbon dioxide. A continuously increasing number of molecular catalysts of these reactions, mostly transition metal complexes, have been proposed, rendering necessary procedures for their rational benchmarking and fueling the quest for leading principles that could inspire the design of improved catalysts. The search of “volcano plots” correlating catalysis kinetics to the stability of the key intermediate is a popular approach to the question in catalysis by surface-active sites, with as foremost example the electrochemical reduction of aqueous proton on metal surfaces. We discussed here for the first time, on theoretical and experimental grounds, the pertinence of such an approach in the field of molecular catalysis. This is the occasion to insist on the virtue of careful mechanism assignments. Particular emphasis is put on the interest of expressing the catalysts’ intrinsic kinetic properties by means of catalytic Tafel plots, which relate kinetics and overpotential. We also underscore that the principle and strategies put forward for the catalytic activation of the above-mentioned small molecules are general as illustrated by catalytic applications out of this particular field. |

