Publication
878
Anal. Chem., 91 (10), 6775-6782, 2019
DOI:10.1021/acs.analchem.9b00851
|
|
|
|
|
|
|
Converting Any Faradaic Current Generated at an Electrode under Potentiostatic Control into a Remote Fluorescence Signal |
|
|
|
Rabia Djoumer, Agnès Anne, Arnaud Chovin*, Christophe Demaille*, Corinne Dejous, Hamida Hallil, Jean-Luc Lachaud
Laboratoire d'Electrochimie Moléculaire, UMR 7591 CNRS, Université Paris Diderot, Sorbonne Paris Cité, 15 rue Jean-Antoine de Baïf, F-75205 Paris Cedex 13, France
Université de Bordeaux, Bordeaux INP, IMS, UMR5218 CNRS, F-33405 Talence, France
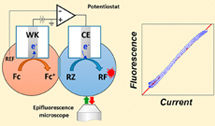
We describe the development of an original faradaic current-to-fluorescence conversion scheme. The proposed instrumental strategy consists of coupling the electrochemical reaction of any species at an electrode under potentiostatic control with the fluorescence emission of a species produced at the counter electrode. In order to experimentally validate this scheme, the fluorogenic species resazurin is chosen as a fluorescent reporter molecule, and its complex reduction mechanism is first studied in unprecedented detail. This kinetic study is carried out by recording simultaneous cyclic voltammograms and voltfluorograms at the same electrode. Numerical simulations are used to account for the experimental current and fluorescence signals, to analyze their degree of correlation, and to decipher their relation to resazurin reduction kinetics. It is then shown that, provided that the reduction of resazurin takes place at a micrometer-sized electrode, the fluorescence emission perfectly tracks the faradaic current. By implementing this ideal configuration at the counter electrode of a potentiostatic setup, it is finally demonstrated that the oxidation reaction of a nonfluorescent species at the working electrode can be quantitatively transduced into simultaneous emission of fluorescence. |

