Publication
895
ChemSusChem., 12 (19), 4500-4505, 2019
DOI:10.1002/cssc.201902040
|
|
|
|
|
|
|
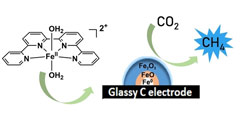
An iron quaterpyridine complex as a precursor for the electrocatalytic reduction of CO2 into methane
|
|
|
|
Claudio Cometto, Lingjing Chen, Daniela Mendoza, Benedikt Lassalle-Kaiser, Tai-Chu Lau, and Marc Robert
Laboratoire d’Electrochimie Moléculaire, UMR 7591 CNRS Universit8 de Paris 75013 Paris (France)
School of Environment and Civil Engineering Dongguan University of Technology Guangdong, 523808 (P.R. China)
Synchrotron SOLEIL L’Orme des Merisiers, Saint-Aubin, 91192, Gif-sur-Yvette (France)
Department of Chemistry and Institute of Molecular Functional Materials, City University of Hong Kong, Hong Kong (P.R. China)
Abstract
A Fe quaterpyridine complex was used as a molecular precursor for the electrochemical reduction of CO2 to CH4 in acetonitrile in the presence of triethanolamine. CH4 was produced with a faradaic yield of approximately 2.1 % at 25 °C and 1 atm pressure of CO2 as reactant. Controlled potential electrolysis coupled to ex situ X‐ray photoelectron spectroscopy and X‐ray absorption spectroscopy of the electrode surface revealed the formation of metallic iron covered by iron oxides as species responsible for catalysis. |

