Publication
902
Chem. Sci., 142 (4), 1006-1010, 2019
DOI:10.1039/C9SC05147C
|
|
|
|
|
|
|
Hydrogen and proton exchange at carbon. Imbalanced transition state and mechanism crossover |
|
|
|
Cyrille Costentin, and Jean-Michel Savéant
Département de Chimie Moléculaire, Université Grenoble-Alpes, CNRS, UMR 5250, 38000 Grenoble, France
Laboratoire d’Electrochimie Moléculaire, Université de Paris, CNRS, UMR 7591, 15 Rue Jean-Antoine de Baïf, F-75013 Paris, France
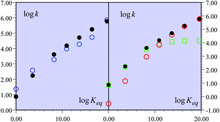
A recent remarkable study of the C–H oxidation of substituted fluorenyl-benzoates together with the transfer of a proton to an internal receiving group by means of electron transfer outer-sphere oxidants, in the noteworthy absence of hydrogen-bonding interactions, is taken as an example to uncover the existence of a mechanism crossover, making the reaction pass from a CPET pathway to a PTET pathway as the driving force of the global reaction decreases. This was also the occasion to stress that considerations based on "imbalanced" or "asynchronous" transition states cannot replace activation/driving force models based on the quantum mechanical treatment of both electrons and transferring protons. |

