Publication
903
ACS Omega, 5 (4), 2015-2026, 2020
DOI:10.1021/acsomega.9b04034
|
|
|
|
|
|
|
Mechanism of Reconstitution/Activation of the Soluble PQQ-Dependent Glucose Dehydrogenase from Acinetobacter calcoaceticus: A Comprehensive Study |
|
|
|
Claire Stines-Chaumeil, François Mavré, Brice Kauffmann, Nicolas Mano, and Benoît Limoges
CNRS, Université de Bordeaux, CRPP, UMR 5031, 115 Avenue Schweitzer, F-33600 Pessac, France
Université de Paris, Laboratoire d’Electrochimie Moléculaire, UMR 7591, CNRS, F-75013 Paris, France
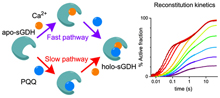
The ability to switch on the activity of an enzyme through its spontaneous reconstitution has proven to be a valuable tool in fundamental studies of enzyme structure/reactivity relationships or in the design of artificial signal transduction systems in bioelectronics, synthetic biology, or bioanalytical applications. In particular, those based on the spontaneous reconstitution/activation of the apo-PQQ-dependent soluble glucose dehydrogenase (sGDH) from Acinetobacter calcoaceticus were widely developed. However, the reconstitution mechanism of sGDH with its two cofactors, i.e., pyrroloquinoline quinone (PQQ) and Ca2+, remains unknown. The objective here is to elucidate this mechanism by stopped-flow kinetics under single-turnover conditions. The reconstitution of sGDH exhibited biphasic kinetics, characteristic of a square reaction scheme associated with two activation pathways. From a complete kinetic analysis, we were able to fully predict the reconstitution dynamics and also to demonstrate that when PQQ first binds to apo-sGDH, it strongly impedes the access of Ca2+ to its enclosed position at the bottom of the enzyme binding site, thereby greatly slowing down the reconstitution rate of sGDH. This slow calcium insertion may purposely be accelerated by providing more flexibility to the Ca2+ binding loop through the specific mutation of the calcium-coordinating P248 proline residue, reducing thus the kinetic barrier to calcium ion insertion. The dynamic nature of the reconstitution process is also supported by the observation of a clear loop shift and a reorganization of the hydrogen-bonding network and van der Waals interactions observed in both active sites of the apo and holo forms, a structural change modulation that was revealed from the refined X-ray structure of apo-sGDH . |

