Publication
913
Inorg. Chem., 59 (16), 11577-11583, 2020
DOI:10.1021/acs.inorgchem.0c01379
|
|
|
|
|
|
|
Electrocatalytic O2 Activation by
Fe Tetrakis(pentafluorophenyl)porphyrin in Acidic Organic Media. Evidence of High-Valent Fe Oxo Species |
|
|
|
Nikolaos Kostopoulos, Célia Achaibou, Jean-Marc Noël, Frédéric Kanoufi, Marc Robert, Claire Fave, and Elodie Anxolabéhère-Mallart
Université de Paris, Laboratoire d’Electrochimie Moléculaire LEM, CNRS, F-75013 Paris, France
Institut Universitaire de France (IUF), F-75005 Paris, France
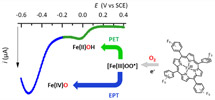
O2 activation under mild conditions remains a weighty challenge for chemists. Herein we report a study of electrochemical O2 reductive activation catalyzed by FeIII(F20TPP)Cl, by means of cyclic voltammetry and UV–vis spectroelectrochemistry in acidic solutions of N,N-dimethylformamide. Two parallel catalytic pathways have been evidenced occurring at different overpotentials. At high overpotential a classical electron–proton (EPT) pathway where protonation of Fe peroxo ultimately leads to the formation of high-valent Fe oxo species dominates. At low overpotential a proton–electron (PET) pathway involving a hydrosuperoxo species has been identified. |

